Pharmatest
Services
Osteogenesis imperfecta
Human Osteogenesis Imperfecta (OI) is a group of genetic disorders that results in heterogeneous clinical symptoms ranging from low bone mass and occasional fractures to perinatal lethality. This heterogeneous disease has been divided into at least 17 subtypes. Most cases of OI are caused by autosomal dominant mutations in the genes encoding type I collagen (COL1A1 and COL1A2) and are classified in subtypes I to IV.
The clinical management of OI has aimed to reduce fracture rates by maximizing bone strength and by minimizing trauma and impact injuries. Pharmacological treatment has been considered as one approach for increasing bone strength and decreasing fracture rates in OI patients.
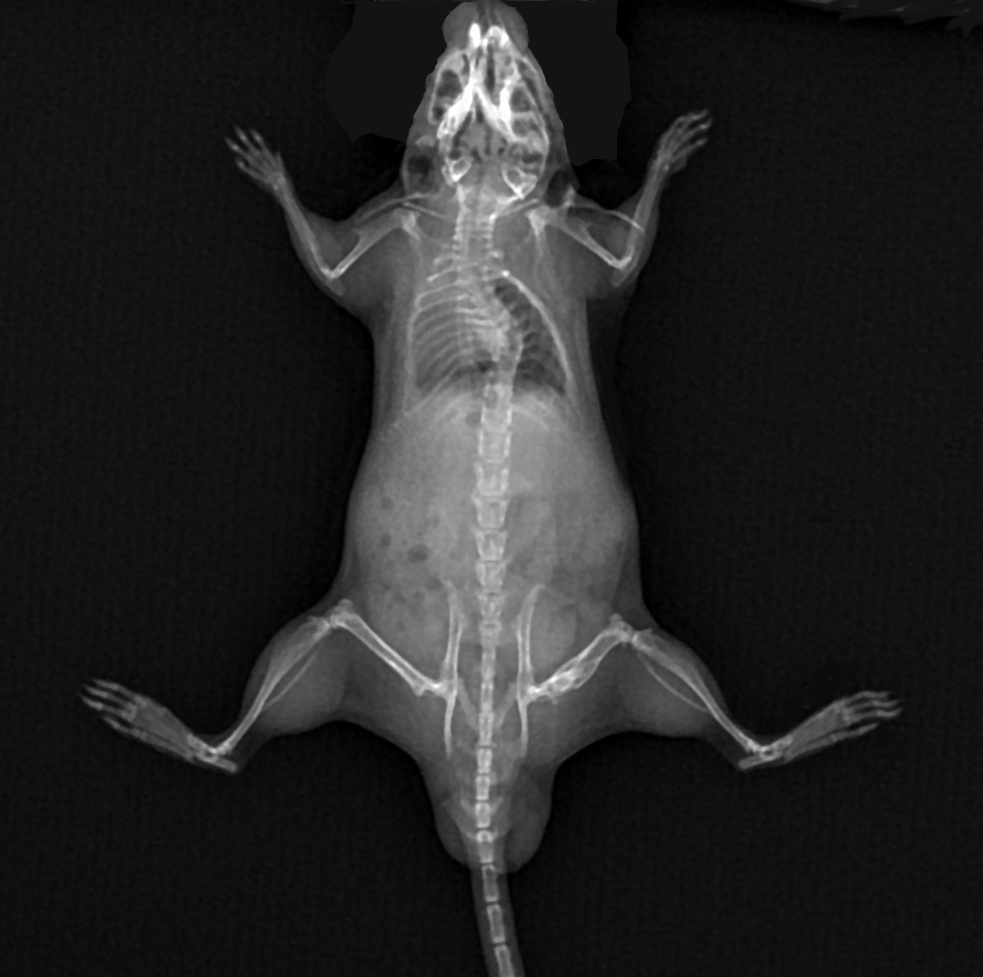
In vivo model for Osteogenesis Imperfecta
Preclinical efficacy studies with animal models of OI provide first-hand information about the effects of new possible therapies before proceeding to clinical trials.
Most OI animal models are gene-modified mice mimicking genetic disorders observed in human patients. Pharmatest offers a mouse model of OI where ‘Amish variant’ mice with a G610C mutation in the Col1a2 gene are used. Heterozygous mice (+/G610C) mimic human type IV OI with a mild-to-moderate severity and exhibit decreased bone mineral density and bone strength.
Contact an Expert